
MANUFACTURING DEFECTS YOU DON’T SEE
This year has been one of heightened awareness regarding battery safety. Smartphone manufacturers now realize that battery fires are real and recalls from the field are enormously expensive. Along with this education comes a deeper realization that battery...
This year has been one of heightened awareness regarding battery safety. Smartphone manufacturers now realize that battery fires are real and recalls from the field are enormously expensive. Along with this education comes a deeper realization that battery quality and the presence of hidden defects are very serious matters.
Today’s post sheds light onto the challenges of battery manufacturing and ensuing defects. These discussions are not useless academic conversations. We see manufacturing defects in batteries especially those sourced from Chinese manufacturers. We observe them more frequently in batteries with high energy density where manufacturing tolerances are challenging.
We analyzed recently a family of batteries from a China-based manufacturer that shall remain unnamed. The battery in question is in serious consideration for a possible release in a smartphone in 2017. Our analysis revealed that the battery was potentially unsafe exhibiting an elevated risk of lithium metal plating; this can lead to electrical shorts. A post-mortem dissection showed regular horizontal bands of lithium metal deposited on the surface of the graphite anode. The scary part was that this battery was quite new….it had only been used for a few days with less than 10 charge-discharge cycles. So what’s happening?

Let’s go back to basics. This previous post recaps the basic structure of a lithium-ion battery. There are two electrodes that face each other with a porous insulating layer in between meant to keep these two electrodes apart. The anode has a coating of carbon (in the form of graphite) on top of a copper layer. The cathode has a coating of a specialized metal oxide, often lithium cobalt oxide abbreviated as LCO.
The design of the battery dictates that the amount of graphite compared to the amount of LCO must be balanced. This balance, which in technical jargon is called the A to C ratio, requires that there is a small amount of excess graphite relative to LCO. Usually, in a good design, there is about 5% more graphite material than LCO.
If, for some reason, there is less graphite than there is LCO, then this creates a condition where excess lithium ions cannot be absorbed by the anode, leading to lithium metal forming on the surface of the graphite….this is called lithium metal plating. As I said before and I will say again, lithium metal is a NO NO. It is a risk for electrical shorts and it is highly flammable in the presence of water vapor or oxygen; both are precursors for poor safety.
In a good manufacturing environment, the graphite anode layer is uniform in thickness and density. In other words, anywhere along the large anode surface, the graphite maintains the proper balance with the cathode layer that sits on the opposite side.
But now imagine a scenario where the manufacturing is not well controlled such that regions of the anode (or the cathode) are not uniform — for example the thickness or the density of graphite particles vary. Now remember that this need not be a huge variation: only a mere 5% change in particle density is sufficient to create an imbalance between the two electrodes. That is precisely what happened in this Chinese battery. The figure below illustrates how these bands are related to the defects in the electrode layer.
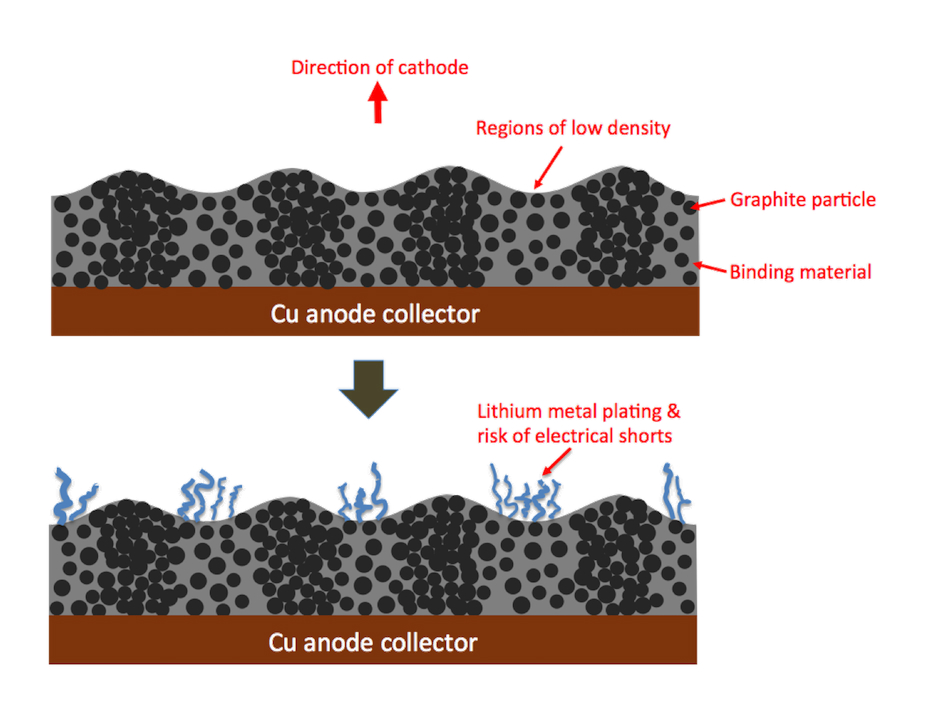
When the manufacturer coated the anode and cathode layers, their machines created small ripples in the density and/or thickness. So when the battery was first powered, lithium metal began to form…and that was what we observed in our laboratory.
So now you will say, “Well, that was a cheap Chinese factory. Surely the more reputable incumbents do not have this problem.” True, but that is not good enough. The economics are not in their favor. I will explain.
The tolerances of manufacturing high-energy-density batteries are becoming very tight. This drives the need for newer state-of-the-art manufacturing equipment, such as machines that can coat the electrodes more uniformly. The cost of equipping new manufacturing facilities therefore increases, driving up the cost of manufacturing these batteries.
Compare this to manufacturing silicon integrated circuits. Moore’s Law makes it possible to spend billions of dollars on manufacturing facilities and yet amortize this cost over a rapidly increasing number of electronic chips. The result is the amortized cost per chip actually goes down. Sadly, there is no equivalent of Moore’s Law in batteries. More expensive manufacturing means more expensive batteries, and consequently, a loss of market share to the low-cost Chinese battery manufacturers. It is no surprise therefore that the reputable incumbents in lithium-ion batteries like LG Chem and Samsung SDI have their sights set on the electric vehicle market where they get to build the power train, not just a battery. This is a big invitation to low-cost and low-quality battery makers in China to continue expanding.
The combination of increasing energy density along with the enormous pricing pressures from second-tier battery manufacturers in China are invariably leading to increased incidence of manufacturing defects — and that is something smartphone and device OEMs ought to be thinking about very seriously.
